The New Frontier of Gene Editing: From Precision Tools to Personalized Therapies
A new generation of programmable gene editing tools is pushing biotechnology toward an era of truly personalized medicine one in which therapies are tailored not only to specific diseases but to individual genetic profiles. As highlighted in a recent npj Biomedical Innovations feature (Nature Portfolio, July 2025), advances in artificial intelligence, data integration, and next-generation CRISPR systems are beginning to merge, transforming how genetic therapies are conceived, tested, and deployed.
The convergence points to a future in which biology itself becomes programmable where digital design and molecular engineering operate as two sides of the same process.
For decades, gene editing relied on painstaking, manual molecular techniques that were limited in precision and accessibility. The arrival of CRISPR-Cas9 changed that trajectory, democratizing gene editing across the life sciences. Yet, as Nature’s November 2025 feature on the growing debate over “CRISPR babies” underscores, the field now stands at a more complex crossroads no longer defined solely by technical progress but by the ethical boundaries of its application.
Scientists agree that while CRISPR’s accuracy and efficiency have improved, its potential use in heritable genome editing raises profound societal questions. The conversation has shifted from whether we can edit the genome to whether and how we should, as precision tools begin to make once-theoretical interventions medically and ethically tangible.
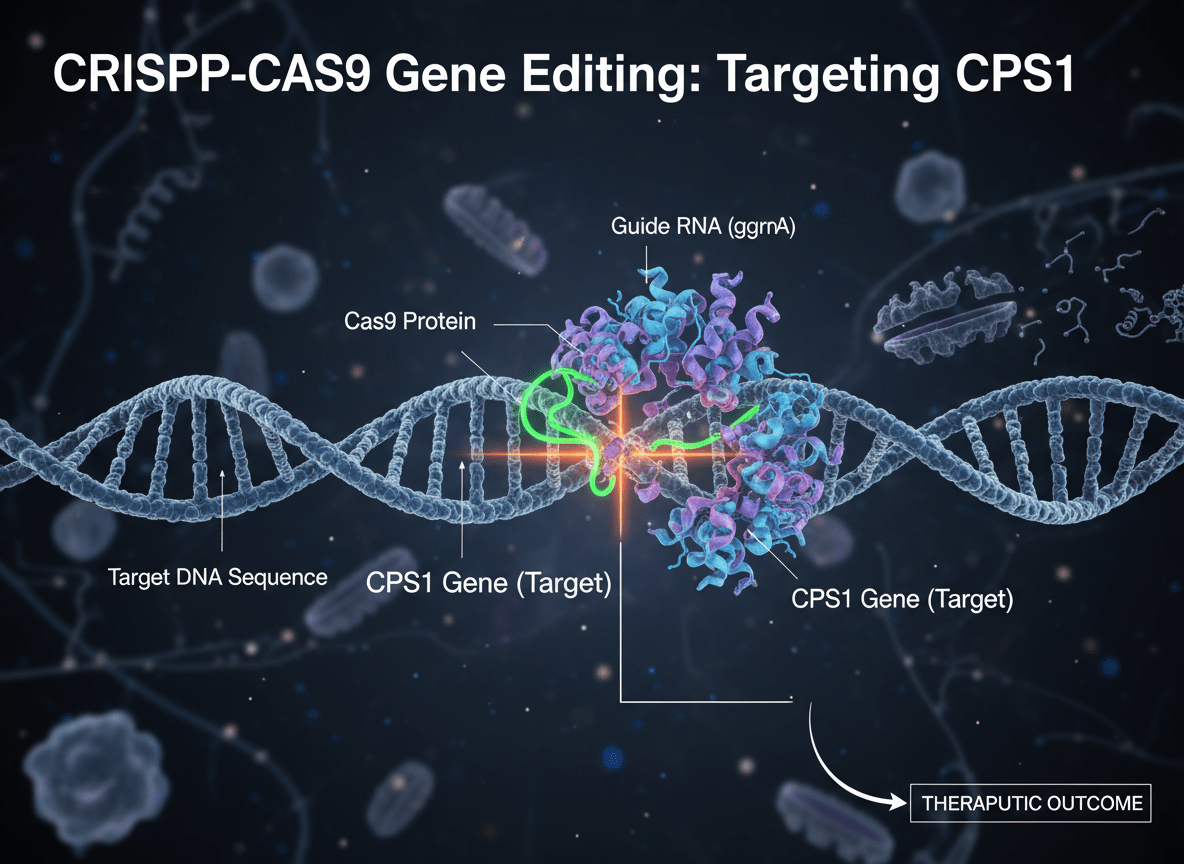
As reported in Nature (May 2025), scientists at the Children’s Hospital of Philadelphia and the University of Pennsylvania designed a base-editing system to correct the single-letter mutation responsible for KJ’s carbamoyl phosphate synthetase 1 (CPS1) deficiency a fatal metabolic disorder that prevents the liver from clearing ammonia from the blood. Delivered through lipid nanoparticles carrying mRNA-encoded editors, the treatment restored his enzyme function and stabilized his metabolism within weeks. Clinicians observed that his ammonia levels remained normal even after common childhood infections, suggesting durable correction. The intervention, approved under an emergency IND by the FDA, demonstrated that a personalized gene therapy could move from design to bedside in under a year.
But as Nature’s October follow-up notes, the field now faces its steepest climb: scaling “N-of-1” medicine to the many. Dozens of rare-disease groups have already approached the Philadelphia team seeking similar interventions. The bottleneck lies not in biology but in manufacturing and regulation every therapy must be synthesized, validated, and quality-checked as if it were a new drug.
U.S. agencies such as ARPA-H are developing modular “GMP pods” to automate bespoke production, while ethicists and regulators weigh new frameworks for patient consent and algorithmic accountability. The success of KJ’s therapy has, in effect, turned one child’s cure into a blueprint for the next generation of personalized gene editing and perhaps, for the democratization of genetic medicine itself.
A new wave of biotech innovators is fusing artificial intelligence with CRISPR to advance precision medicine. Scribe Therapeutics’ AI-powered DeepXE platform designs novel, efficient CRISPR enzymes with fewer off-target effects. Beam Therapeutics uses computational modelling to refine base-editing tools for single-letter DNA changes, while Profluent Bio applies large language models to create new genome editors, including its open-source OpenCRISPR-1. Together, these efforts show how AI is transforming gene editing from an analytical tool into a generative engine for next-gen therapies.
Academic laboratories are likewise building adaptive CRISPR libraries that evolve based on live feedback from cell systems, effectively closing the loop between data and design. Together, these advances point toward a future where precision gene editing becomes not a static, one-time intervention, but a continuously improving system a self-learning framework that redefines the speed, accuracy, and scope of genetic medicine.
A crucial enabler of this shift is the rise of AI-integrated database and computing infrastructure. Tools such as EDB Postgres AI, Nvidia’s BioNeMo, and Amazon’s Genomics Cloud are becoming the digital backbone for modern life sciences. By embedding multi-omic datasets within intelligent database architectures, these platforms enable researchers to perform complex vector searches across genetic, proteomic, and clinical datasets simultaneously. This integration allows predictive algorithms to correlate gene editing with real-world phenotypic outcomes effectively transforming static genomic repositories into dynamic, learning environments.
As artificial intelligence becomes embedded in gene-editing research, regulators are rethinking oversight frameworks. The U.S. Food and Drug Administration’s 2025 draft guidance on AI in drug and biological development emphasizes “risk-based credibility” requiring explainable, auditable algorithms throughout therapeutic design and validation. In contrast to traditional lab workflows, AI-driven biology is compressing timelines from years to weeks, prompting regulators to prioritize traceability and model accountability.
When unveiling the Evo 2 platform, Stanford engineers captured the magnitude of their advance, noting that “Trained on a dataset that includes all known living species – and a few extinct ones – Evo 2 can predict the form and function of proteins in the DNA of all domains of life and run experiments in a fraction of the time it would take a traditional lab”.
Clinical translation remains a bottleneck. Demonstrating reproducibility across diverse patient populations will be essential before personalized gene editing moves from concept to clinic. To bridge this gap, companies are increasingly turning to real-world evidence (RWE) networks that track patient outcomes longitudinally. Such data streams can validate whether algorithmically designed edits achieve their intended therapeutic effects over time, providing a feedback mechanism for both clinical safety and commercial viability.
While the science of gene editing advances rapidly, questions around reimbursement and equitable access are emerging in parallel. Explore our latest coverage on reimbursement policy and pricing frameworks.
For media inquiries or to share perspectives on access and evidence policy, email editor@synopulse.com.